As the Food and Drug Administration (FDA) released its authorization for the first coronavirus vaccine manufactured by Pfizer, many Americans see a glimpse of hope on the horizon for the days to return to work, school, and to resume life before the pandemic. The promise of effective vaccines arrived at a particularly pivotal moment as the coronavirus infection rates reach new record-breaking numbers in many parts of the United States. With many states recently mandating a safer-at-home order or new restrictions to curb the alarming surge of cases and hospitalization, the promising news of vaccines finally offers a rare moment of hope in the midst of deadly pandemic.
While there are many questions in terms of the safety and distribution timeline of the vaccine, many parents and students across the country wonder if schools can reopen their campuses for in-person classes soon. Although clinical trials for the vaccine have been tested successfully on adults, many health experts are unsure whether they will be safe for children since trials have just begun.
“I think it’s way too early to say whether the school will require or recommend vaccinations for COVID-19 once they become widely available … Once there is good data about the long-term safety of administering the COVID-19 vaccines on minors, it’s possible that the State Legislature will add COVID-19 to the list,” says Harvard-Westlake Director of Communications Ari Engelberg.
According to an article by Denise Grady of New York Times, “the new data from Moderna [another vaccine manufacturer] show its study of 30,000 people has met the scientific criteria needed to determine whether the vaccine works. The findings from the full set of data found the vaccine to be 94.5 percent effective” Another significant finding is that the vaccine was 100 % effective at preventing severe cases of the disease that can lead to critical conditions.
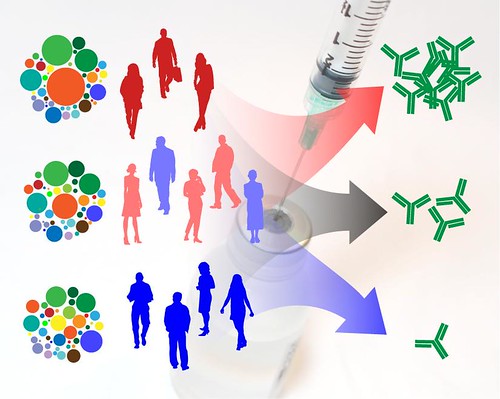
While vaccines can take many different forms, they typically contain an inactivated virus or weakened live virus to ensure that they don’t harm the body by replicating. Vaccines also contain genetic molecules called messenger RNA (or mRNA) which stimulates the immune system and will result in long lasting protection against the virus as the body produces antibodies. After the vaccines are designed, they must go through a preliminary stage followed by three stages of testing. Both Pfizer and Moderna, who also applied for the emergency use of vaccines for FDA approval, have developed vaccines that are similar in that they both use mRNA and that they coax our cells to make the same viral protein, called spike proteins.
So, now that Pfizer vaccine is approved, who will get vaccinated first, and when? While different states will implement their own distribution plans, David Leonhardt of New York Times offers a preliminary timeline of when Americans can expect to get vaccinated. As recommended by the Centers for Disease Control and Prevention (CDC), healthcare workers and nursing home residents are likely to be the first to get vaccinated by the end of December.
The next set of groups in January will include people working at doctor’s offices, hospitals and pharmacies as well as specialty clinics, but about a large number of vaccines will also have to be used as a second dose for those who received shots earlier since both Pfizer and Moderna vaccines require a second dose to be effective. By February and March, it will likely be people over 65 or people with medical conditions as well as essential workers in education, food or transportation, and finally by April through June, healthy, nonessential workers younger than 65 will begin to get vaccinated.
It is projected that the majority of Americans could be vaccinated by early summer; the experts hope that with the effective distribution of vaccines and the flattening of the curve, the end of the unprecedented pandemic may be near. While we continue to see the surging infection rates breaking new records since the Thanksgiving holiday, the news of vaccine approval is just in time to offer the holiday spirit we need heading into 2021.